“Gut health” has become one of the most searched wellness topics on the internet — and for good reason. In the last decade, microbiome science has shifted from a niche area of research to one of the most active and consequential fields in medicine. We now understand that the community of trillions of microorganisms living in your digestive tract is not incidental to your health — it is foundational to it.
But with the surge in interest has come an equally significant surge in misinformation. Probiotic supplements are marketed for everything from weight loss to anxiety relief, while the actual science tells a more nuanced story: some interventions are well-supported, others have weak evidence, and the context — which strains, for which conditions, in which people — matters enormously.
This post is the guide I wish more patients had before spending money on supplements. It covers what the gut microbiome actually is, how it affects your body systems, what probiotics can genuinely do, how to choose and use them wisely, and — critically — why this topic deserves particular attention for women navigating the specific stressors and health conditions that disproportionately affect our community.
What Is the Gut Microbiome?
The gut microbiome refers to the collective community of microorganisms — bacteria, viruses, fungi, archaea, and other microbes — that inhabit your gastrointestinal tract, from your mouth to your colon. The colon alone houses an estimated 38 trillion microbial cells, with the highest concentration per surface area in a region called the cecum, at the junction of the small and large intestines.
This is not a passive passenger system. The gut microbiome functions as a metabolically active organ — producing enzymes, synthesizing vitamins, generating signaling molecules, training your immune system, and communicating bidirectionally with your brain. It weighs approximately 1–2 kilograms and contains roughly 1,000 distinct bacterial species, collectively harboring far more genetic information than the human genome itself.
How does your microbiome develop?
You first encounter your microbiome at birth. Vaginal delivery exposes the newborn to the mother’s vaginal and fecal microbiota — establishing the first significant colonization. Cesarean-born infants have a meaningfully different early microbiome composition, with lower Lactobacillus and higher Staphylococcus initially — though the gap narrows significantly in the first year of life. Breastfeeding contributes further, delivering Bifidobacterium species and human milk oligosaccharides that serve as prebiotics feeding them.
Over the first 2–3 years, a child’s microbiome becomes increasingly diverse and stabilizes into a relatively adult-like composition. From there, it continues to be shaped throughout life by diet, medications, stress, sleep, environment, and illness.
What the Gut Microbiome Does: The Systems It Governs
The breadth of what your gut microbiome influences is one of the most striking developments in modern medicine. Here are the major systems it affects, with the current level of evidence behind each.
Immune Function
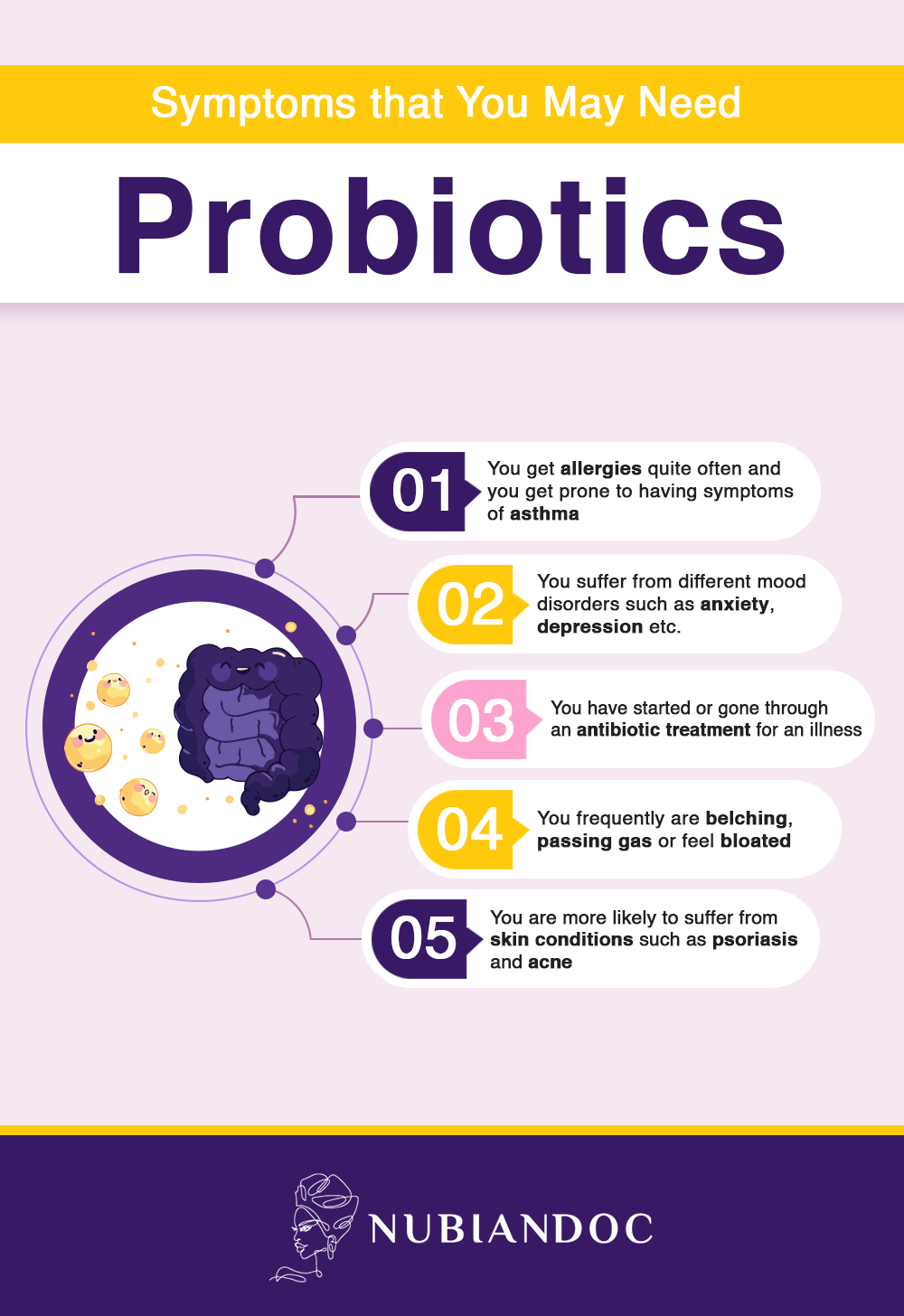
Approximately 70% of the immune system is housed in the gut. The gut microbiome actively trains and calibrates immune responses — teaching immune cells to distinguish between harmless environmental exposures and genuine threats, modulating inflammatory responses, and producing compounds that enhance immune barrier integrity. Dysbiosis (microbial imbalance) is increasingly recognized as a contributor to autoimmune conditions, allergies, and chronic low-grade inflammation that underpins most major chronic diseases.
Digestive Function and Short-Chain Fatty Acids
Gut bacteria break down dietary fiber that your own digestive enzymes cannot process — producing short-chain fatty acids (SCFAs) including butyrate, propionate, and acetate as byproducts. These are not waste products. They are metabolically critical: butyrate is the primary fuel source for colonocytes (the cells lining your colon), and propionate and acetate travel via the bloodstream to influence liver metabolism, blood sugar regulation, appetite signaling, and systemic inflammation. A fiber-depleted diet doesn’t just reduce fiber intake — it starves the microbial communities that produce these essential compounds.
The Gut-Brain Axis
The gut and brain are in constant bidirectional communication via the vagus nerve, the enteric nervous system, immune signaling, and gut-derived hormones and neurotransmitters. Roughly 90% of the body’s serotonin is produced in the gut. Gut bacteria directly influence the production of GABA, dopamine precursors, and other neuroactive compounds.
A 2025 meta-analysis published in Healthcare synthesized evidence from randomized controlled trials on gut microbiome-targeted interventions for mental health in women across hormonal life stages — including the menstrual cycle, pregnancy, postpartum, and menopause — and found meaningful reductions in depression and anxiety symptoms. Probiotics also showed significant improvements in psychological symptoms of menopause in a 2025 systematic review. This is the gut-brain axis in clinical action.
A 2025 Scientific Reports study found that high levels of perceived stress were associated with lower gut microbial alpha diversity — the richness and evenness of species — and that different stress domains (psychological, physiological, and life event-based) affected distinct microbial pathways. Chronic stress doesn’t just affect your mood. It restructures your microbiome.
Metabolic Health: Insulin, Weight, and Diabetes Risk
The composition of your gut microbiome is now recognized as a meaningful contributor to metabolic health. Microbial diversity is inversely associated with obesity, insulin resistance, and type 2 diabetes risk. Specific bacterial genera produce SCFAs and metabolites that improve insulin sensitivity and reduce hepatic fat accumulation. The 2025 sugar study referenced in our post on how added sugar affects your health confirmed that dietary sugars — glucose, fructose, and sucrose — deplete SCFA-producing bacteria and impair gut barrier integrity, creating pathways from dietary pattern to systemic metabolic disease via the microbiome.
Cardiovascular Health
Gut bacteria metabolize dietary components — particularly choline and L-carnitine from red meat — into trimethylamine N-oxide (TMAO), a compound associated with increased cardiovascular risk. Conversely, certain bacterial strains produce metabolites that lower blood pressure and reduce LDL cholesterol by breaking down bile acids. The cardiovascular implications of gut microbiome composition are an active and rapidly growing area of research.
Colorectal Cancer Risk
Gut microbial composition is associated with colorectal cancer risk. Research in Black women specifically — who face a 48% greater risk of colorectal cancer diagnosis than white women even after controlling for known risk factors — has found differences in microbial composition that may be a contributing factor. Microbial diversity, fiber intake, and the ratio of protective to potentially pro-inflammatory bacterial species are all implicated.
The Microbiome, Stress, and Why This Matters for You
This is where the science becomes most directly relevant to the lived experience of Black women.
A 2024 study published in BMC Women’s Health examined the relationships among psychosocial stress, diet, and gut microbiome composition in Black and white women and found that life stress variables were significantly correlated with multiple gut microbial genera — including lower levels of beneficial Bifidobacterium and Blautia with higher stress. A 2025 PNAS-supported analysis confirmed that chronic stress — including from racism, discrimination, economic insecurity, and sleep disruption — alters gut microbial composition in ways that increase inflammatory burden and metabolic disease risk.
A 2025 meta-analysis of African American gut microbiome datasets found significantly lower microbial alpha diversity in African Americans compared to non-African Americans — with ethnicity demonstrating a stronger effect on microbiome variation than diet, age, sex, or BMI individually. Lower microbial diversity is consistently associated with higher risk for obesity, diabetes, and chronic inflammatory disease.
This is not simply a genetic story. The same PNAS analysis notes that the factors driving these microbial differences — high-fat, low-fiber diets in food-insecure environments; higher chronic stress burden; reduced access to outdoor green space; disrupted sleep from shift work — are structural, not individual. Understanding the gut microbiome as a biological pathway through which social and environmental adversity becomes embodied disease is one of the most important frameworks emerging from microbiome science.
Caring for your gut is not a luxury. It is, for many women, a meaningful form of health equity practice.
What Are Probiotics — And What Is the Evidence?
Probiotics are defined by the International Scientific Association for Probiotics and Prebiotics as “living microorganisms that, when administered in adequate amounts, confer a health benefit on the host.” This definition is important: not every product marketed as a probiotic meets this threshold.
A 2026 BMC Medicine systematic review and meta-analysis of randomized controlled trials found that probiotic supplementation modestly but significantly increased gut microbial diversity in healthy populations — with effects most pronounced for multi-strain formulations and when taken for longer durations. Effects were transient in some studies, returning to baseline after supplementation stopped, underscoring that dietary patterns are the more durable driver of microbiome composition.
Here is where the evidence is strongest — and where it remains limited:
Well-Supported by Evidence
Antibiotic-associated diarrhea: This is one of the most consistently demonstrated applications of probiotics. Antibiotics disrupt the gut microbiome indiscriminately — killing beneficial bacteria alongside pathogens — often causing diarrhea as a consequence. Multiple meta-analyses show that Lactobacillus rhamnosus GG and Saccharomyces boulardii significantly reduce the incidence and duration of antibiotic-associated diarrhea. If you are prescribed antibiotics, taking a probiotic simultaneously (separated by 2 hours from the antibiotic dose) is a clinically reasonable strategy.
IBS with constipation and bloating: Specific probiotic strains — particularly multi-strain Lactobacillus and Bifidobacterium combinations — have demonstrated efficacy in improving stool frequency, consistency, and bloating in people with IBS and functional constipation. The effect is strain-specific; broad claims that “any probiotic helps IBS” are not supported. See our post on constipation causes and treatment for more detail.
Vaginal microbiome and UTIs: The vaginal microbiome — dominated by Lactobacillus species in most women — plays a protective role against bacterial vaginosis and recurrent urinary tract infections. Emerging 2024 research confirms that oral or vaginal probiotic supplementation with Lactobacillus strains may help restore vaginal microbiome balance and reduce recurrence of these conditions in women who are prone to them.
Mental health across hormonal transitions: As noted above, the 2025 systematic review found meaningful improvements in depression and anxiety symptoms in women using gut microbiome-targeted interventions during hormonal transitions. Probiotics containing Bifidobacterium longum and Lactobacillus helveticus have the most consistent data for mood outcomes.
PCOS and metabolic outcomes: Probiotic supplementation has been shown in multiple trials to improve insulin sensitivity, reduce androgen levels, and support more regular menstrual cycles in women with PCOS — consistent with its role in metabolic regulation discussed in our intermittent fasting post.
Promising But Not Yet Definitive
Weight management: Some studies show that specific probiotic strains influence weight and body composition, but results are inconsistent across trials. Probiotics are not a weight loss intervention. Their role is supportive of metabolic health, not a primary driver of weight change.
Eczema and allergic conditions: Evidence suggests early-life probiotic exposure may reduce atopic dermatitis risk, and some adult studies show benefit for eczema severity — but strain specificity and timing matter significantly.
Immune resilience: Probiotic supplementation during upper respiratory illness season appears to reduce duration and severity of infections in some populations, but the data is mixed.
Where Evidence Is Weak or Absent
Probiotics for autism, neurological disease, or cancer prevention remain early-stage and speculative. The marketing has significantly outpaced the clinical evidence in these areas.

Probiotics and Bloating: What’s Actually Happening
Bloating is one of the most common digestive complaints — and one of the most marketed probiotic indications. Here is what is actually going on physiologically.
Bloating typically results from one or more of the following:
- Excess gas production from fermentation of undigested carbohydrates by gut bacteria — particularly FODMAPs (fermentable sugars in beans, wheat, onions, some fruits, dairy)
- Small intestinal bacterial overgrowth (SIBO) — an abnormal proliferation of bacteria in the small intestine where bacterial density should be low
- Visceral hypersensitivity — heightened perception of normal gut distension, common in IBS
- Slowed motility — gas and content moving too slowly through the gut
Probiotics may help bloating by shifting the microbial community toward less gas-producing strains, improving gut motility, or reducing visceral sensitivity in IBS. The effect is real in some people for some causes of bloating — but it is not universal, and it is not the same as treating the underlying cause. If bloating is severe, persistent, or associated with significant pain, weight loss, or blood in stool, medical evaluation is essential — not a probiotic supplement.
Food Sources vs. Supplements: Which Is Better?
For most healthy people, food-based sources of probiotics are the preferred approach — they come packaged with other nutritional benefits, are generally well-tolerated, and support the broader dietary patterns that sustain microbial diversity.
Best food sources of probiotics:
| Food | Key Strains | Notes |
|---|---|---|
| Yogurt (with live cultures) | Lactobacillus, Bifidobacterium | Look for “live and active cultures” on label |
| Kefir | Multiple Lactobacillus and yeast strains | Higher diversity than yogurt; tangy flavor |
| Kimchi | Lactobacillus plantarum, others | Also high in fiber and antioxidants |
| Sauerkraut (unpasteurized) | Lactobacillus species | Pasteurized versions have no live cultures |
| Miso | Aspergillus oryzae, Lactobacillus | Use in soups and dressings without boiling |
| Tempeh | Rhizopus oligosporus | Also an excellent plant protein source |
| Kombucha | Acetobacter, Lactobacillus | Variable sugar and alcohol content; choose lower-sugar varieties |

Note on dairy-based probiotics and lactose intolerance: Yogurt and kefir are among the most tolerated dairy products for people with lactose intolerance because the live cultures partially pre-digest the lactose. As discussed in our dairy post, these are specifically recommended options for Black women navigating lactose intolerance without forgoing the nutritional benefits of fermented dairy.
When supplements make sense:
- During and after a course of antibiotics
- When you have a specific, evidence-supported indication (IBS-C, recurrent BV, antibiotic-associated diarrhea)
- When food-based sources are not accessible or tolerated
- During travel (where gut flora disruption from new food environments is common)
Choosing a Probiotic Supplement: What to Look For
If you are choosing a supplement, here is what actually matters:
CFU count: CFU stands for colony-forming units — a measure of viable bacterial cells. Most clinical trials use doses between 1 billion and 100 billion CFU. Higher is not automatically better. Match the dose to the clinical evidence for your specific indication.
Strain specificity: Benefits are strain-specific, not genus-wide. Lactobacillus acidophilus and Lactobacillus rhamnosus GG have different evidence profiles and different applications. Look for the full strain name on the label, not just the genus.
Multi-strain vs. single-strain: For general gut health and microbiome diversity, multi-strain formulations tend to show stronger effects than single-strain. For specific applications (e.g., antibiotic-associated diarrhea), single well-studied strains may be more appropriate.
Survivability and storage: Probiotics must survive the acidic environment of the stomach to reach the colon alive. Look for enteric-coated capsules or evidence of gastric stability. Follow storage instructions — some require refrigeration, others are shelf-stable. Always check the expiration date.
Third-party testing: As with all supplements, the FDA does not review probiotic supplements before they reach market. Look for products with NSF International or USP verification seals confirming purity and potency.
Prebiotics: The Fuel That Makes Probiotics Work
No discussion of the microbiome is complete without addressing prebiotics. While probiotics introduce beneficial microorganisms, prebiotics feed the beneficial organisms already present in your gut. They are indigestible fibers that selectively support the growth of health-promoting bacterial species.
Key prebiotic foods:
- Garlic, onions, leeks, shallots
- Asparagus, Jerusalem artichokes
- Oats, barley, rye
- Unripe bananas
- Chicory root (the most concentrated natural inulin source)
- Flaxseed and chia seed
- Legumes (beans, lentils, chickpeas)
The combination of probiotics plus prebiotics is sometimes called a synbiotic approach — and this combination is what the emerging evidence most strongly supports for durable microbiome support. A probiotic supplement without adequate dietary fiber to feed the organisms is a significantly less effective strategy.

What Disrupts the Gut Microbiome
Understanding what harms your microbiome is as important as knowing how to support it.
Antibiotics are the most significant acute disruptor. They are sometimes medically necessary — but should only be used when genuinely indicated, and always followed by intentional microbiome restoration through diet and, when appropriate, probiotics.
Ultra-processed foods and added sugar deplete microbial diversity, reduce SCFA production, and promote inflammatory bacterial populations. The dietary pattern most consistently associated with a healthy, diverse microbiome is high in plant diversity, fiber, fermented foods, and whole foods — and low in processed ingredients.
Chronic psychological stress — as described above — is now confirmed to alter microbial diversity and composition in measurable ways. Stress management is microbiome management. Our post on how social connections affect your health addresses the physiological consequences of chronic stress on gut health and systemic inflammation.
Insufficient sleep disrupts the gut circadian rhythm — the microbiome has its own internal clock — and alters microbial composition in ways that increase metabolic risk.
Sedentary lifestyle: Regular physical activity is associated with greater gut microbial diversity and higher levels of SCFA-producing bacteria. The mechanisms are partly mechanical (colonic motility) and partly metabolic. See our exercise post for more on how movement benefits far more than what most people realize.
Overuse of laxatives and non-essential medications: Multiple classes of common medications — PPIs (proton pump inhibitors), metformin, NSAIDs — alter gut microbial composition with regular use. This doesn’t mean avoiding necessary medications; it means being intentional about what you take and why.
The Bottom Line
Your gut microbiome is one of the most powerful determinants of your health — influencing immune function, metabolic health, mental health, cardiovascular risk, and more. It is shaped by everything from how you were born to what you eat, how much you sleep, how stressed you are, and what medications you take.
Probiotics are a genuine clinical tool — particularly for antibiotic-associated diarrhea, IBS, vaginal microbiome health, and increasingly for mental health outcomes during hormonal transitions in women. They are most effective when strain-specific, paired with prebiotic-rich dietary patterns, and used as part of a broader approach to gut health rather than as a standalone supplement.
The most evidence-supported strategy for microbiome health is not a pill. It is dietary diversity — specifically, eating 30 or more different plant foods per week across vegetables, fruits, whole grains, legumes, nuts, and seeds — combined with consistent fermented food intake, adequate sleep, regular movement, and chronic stress management.
For those navigating the structural stressors that disproportionately affect gut microbial diversity in our community, this information is not just wellness advice. It is a clinically grounded argument for treating your gut health as a priority — because the science now confirms it has profound downstream effects on the chronic conditions our community faces at the highest rates.
For more on how nutrition, lifestyle, and hormones connect to your gut and overall health, read our posts on why constipation happens and what actually helps, dairy, lactose intolerance, and gut health, how sugar affects your gut microbiome, vitamin supplementation and the gut, and maintaining a healthy weight through gut-supportive eating.
References:
- Chen PW. The Latest Research on Probiotics: Nourishing Health or Pathways to Disease Resolution. Nutrients. 2024;17(1):19.
- Gut Microbiota for Health. Key Advances in the Gut Microbiome During 2024. January 2025.
- Liaquat M et al. The gut microbiota in menopause: Is there a role for prebiotic and probiotic solutions? Ther Adv Endocrinol Metab. 2025.
- BMC Medicine. Effect of probiotic supplementation on gut microbiota diversity in healthy populations: systematic review and meta-analysis of RCTs. January 2026.
- Guerreiro CS et al. Efficacy of Gut Microbiome-Targeted Interventions on Mental Health Symptoms in Women. Healthcare. 2025;13(22):2851.
- Scientific Reports. Biological, environmental, and psychological stress and the human gut microbiome in healthy adults. 2025.
- BMC Women’s Health. Psychosocial stress, dietary intake and gut microbiota in Black and White women, Birmingham, AL. 2024.
- Brewster R et al. Meta-Analysis of the Gut Microbiome: An African American Representation. IJERPH. 2025;22(10):1591.
- Dahl WJ et al. The human gut microbiome and health inequities. PNAS. 2021.
- Siddiqui R et al. The Gut Microbiome and Female Health. Biology. 2022;11(11):1683.
- NIH NCCIH. Probiotics: What You Need to Know.
- Healthline. How Your Gut Microbiome Impacts Your Health.

